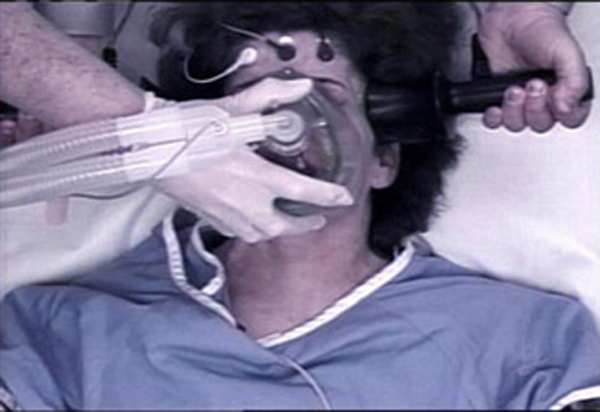
FDA: Electroshock has risks but is useful to combat severe
$ 162.99
-
By A Mystery Man Writer
-
-
5(170)

Product Description

Can Your Risk Management Prevent a Recall? — Exeed
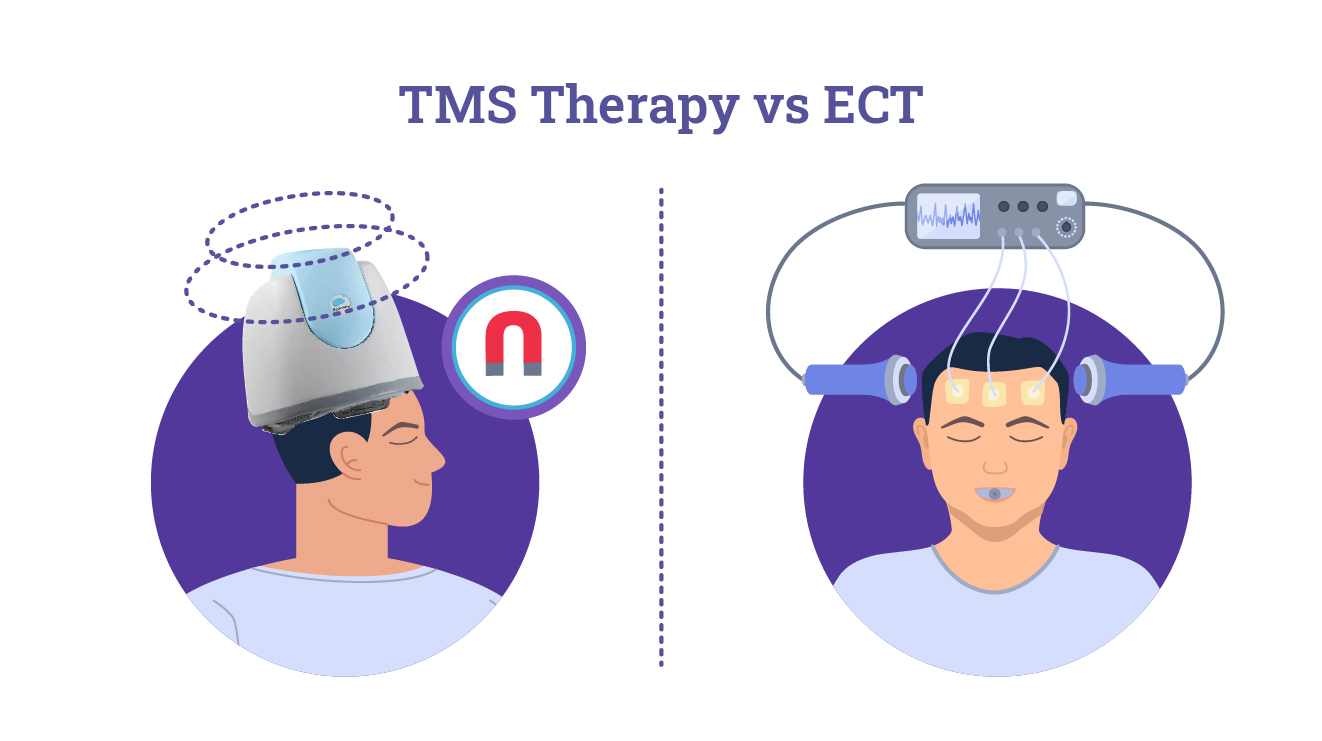
TMS Therapy VS ECT: What's The Difference And When Should ECT NOT Be Used?

FDA considers ban on electric shock conditioning for autistic patients, US news
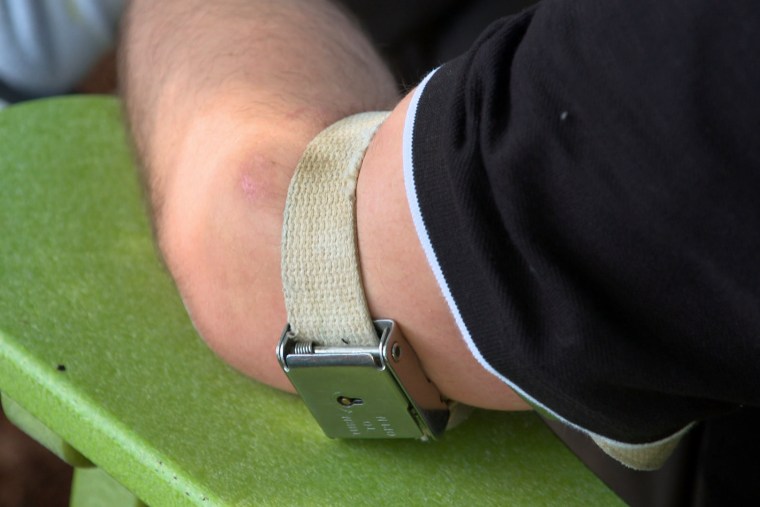
A decades-long fight over an electric shock treatment led to an FDA ban. But the fight is far from over.
FDA panel deems electroshock devices high risk

FDA: Electroshock has risks but is useful, News

Controversial skin shocks used at Mass. school draw FDA look

FDA ban on electric shock treatment at Massachusetts school overturned

FDA Clears Remote Electrical Neuromodulation Device for Use in Younger Patients

A decades-long fight over an electric shock treatment led to an FDA ban. But the fight is far from over.

School Shocks Students With Disabilities. The FDA Is Moving To Ban The Practice : NPR
Debunked! 4 myths about electroconvulsive therapy for depression, Brain, Mental Health

FDA to ban use of electric shock devices to treat children